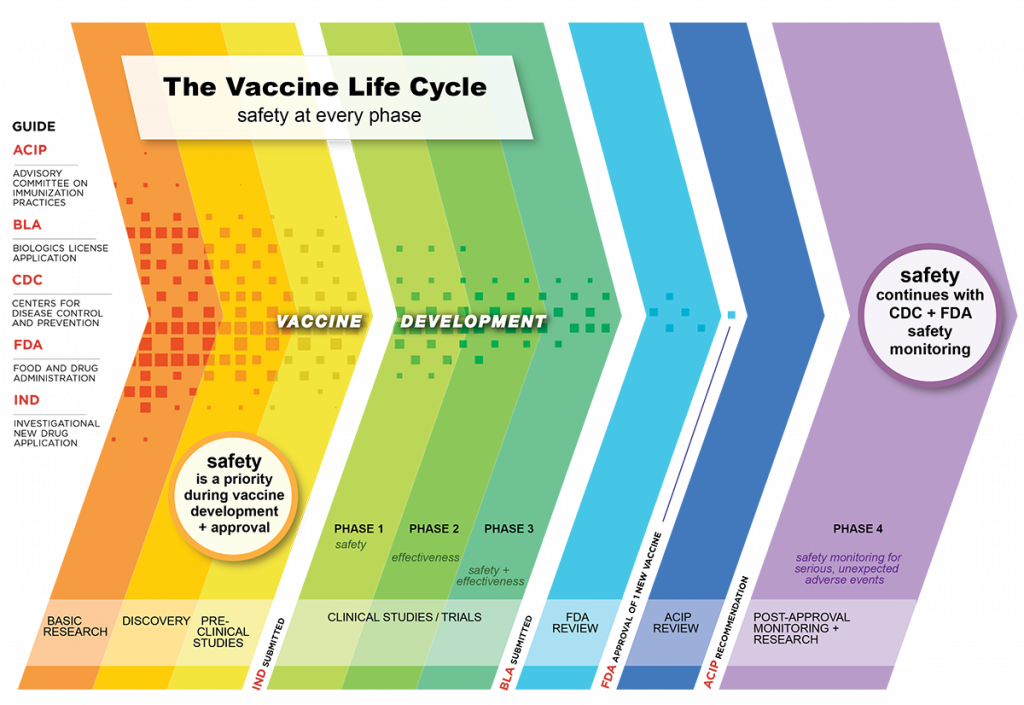
The story of vaccines today – Development and Life cycle
The vaccines’ story didn’t begin with the primary vaccine–Edward Jenner’s use of material from cowpox pustules to supply protection against smallpox. Adequately, it creates with the long history of infectious disease in humans, and especially with early uses of smallpox material to provide immunity to that disease.
Evidence exists that the Chinese employed smallpox inoculation (or Variolation, intrinsically use of smallpox material, was called) as early as 1000 CE. It had practiced in Africa and Turkey before it spread to Europe and, therefore, the Americas.
Edward Jenner’s innovations, begun together with his successful 1796 use of cowpox material to make immunity to smallpox, quickly made the practice widespread. His method underwent medical and technological changes over the subsequent 200 years and eventually resulted in eradicating smallpox.
Edward Jenner’s innovations, begun together with his successful 1796 use of cowpox material to make immunity to smallpox, quickly made the practice widespread. His technique underwent medical and technological changes over the subsequent 200 years and eventually resulted in eradicating smallpox.
Louis Pasteur’s 1885 rabies vaccine was after form an impression on human disease. And then, at the dawn of microbiology, developments swiftly followed. Antitoxins and vaccines in contradiction of diphtheria, tetanus, anthrax, cholera, plague, typhoid, tuberculosis, and more were established through the 1930s.
The middle of the 20th century was a lively time for vaccine research and development. Methods for growing viruses within the laboratory led to rapid discoveries and innovations, including vaccines for polio. Researchers targeted other common childhood diseases like measles, mumps, rubella, and vaccines for these diseases significantly reduced the disease burden.
Innovative techniques now drive vaccine research, with recombinant deoxyribonucleic acid technology and new delivery techniques leading scientists in new directions. Disease targets have expanded, and a few vaccine research is starting to specialize in non-infectious conditions like addiction and allergies.
More than the science following vaccines, these timelines shield cultural aspects of vaccination also, from the first harassment of smallpox variolation (see the intimidation of a prominent minister described within the 1721 Boston Smallpox Epidemic entry) to the establishment of vaccination mandates, to the effect of war and social unrest on vaccine-preventable diseases. Jenner , Pasteur , and Maurice Hilleman, pioneers in vaccine development, receive particular attention also.
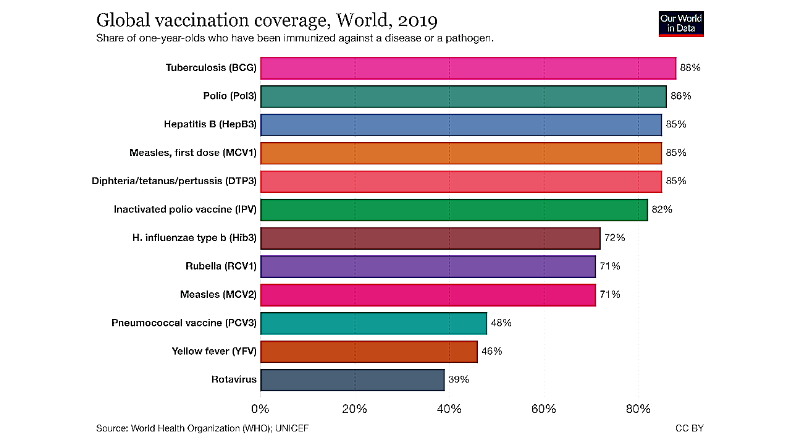
Global vaccination coverage 2019
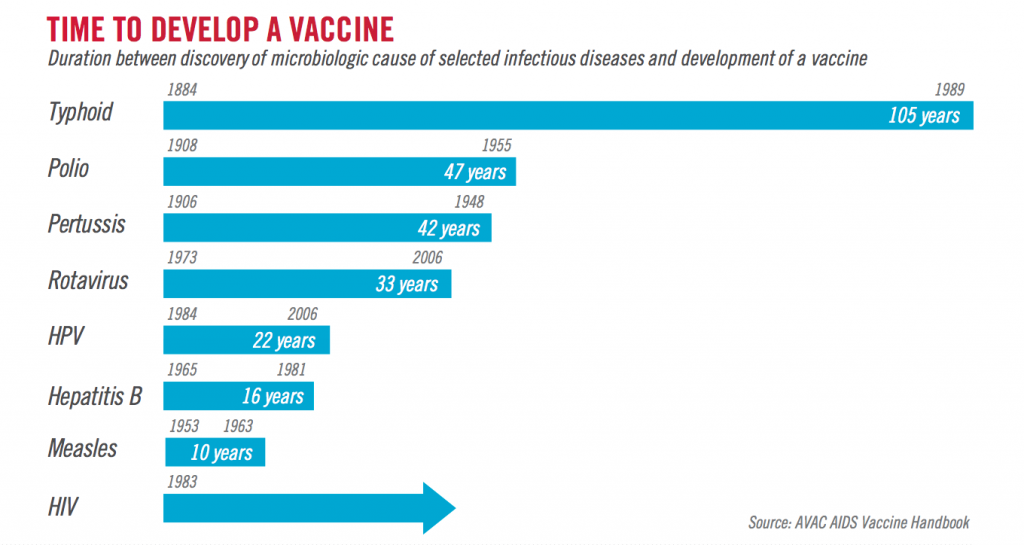
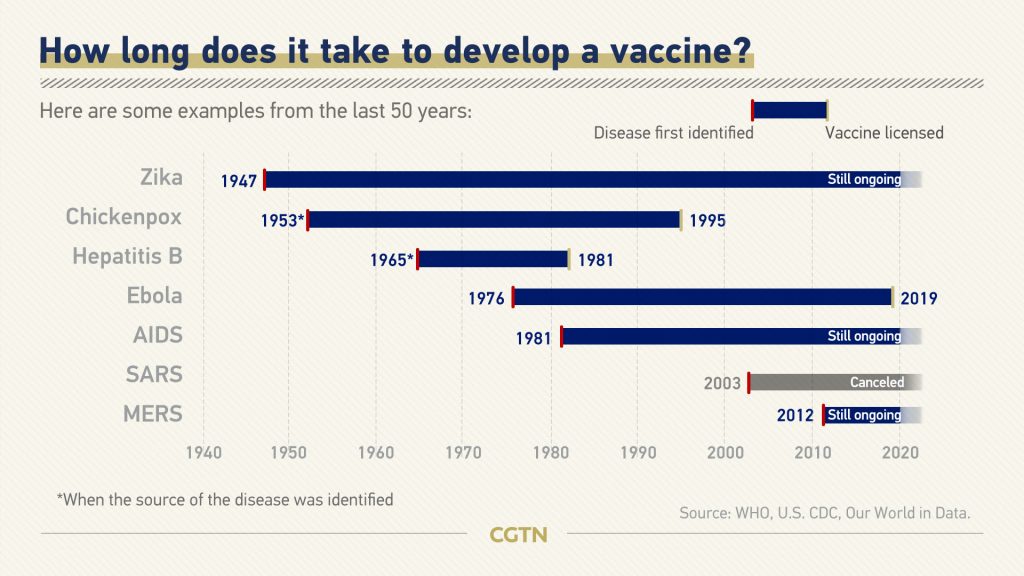
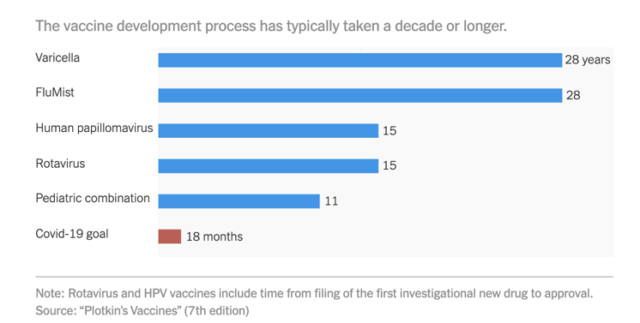
The story of vaccines – How long it took to develop
Jennifer Pancorbo: There are two ways to interpret this question. Do you mean actually manufacturing a vaccine that is already created? Or do you mean designing a new vaccine?
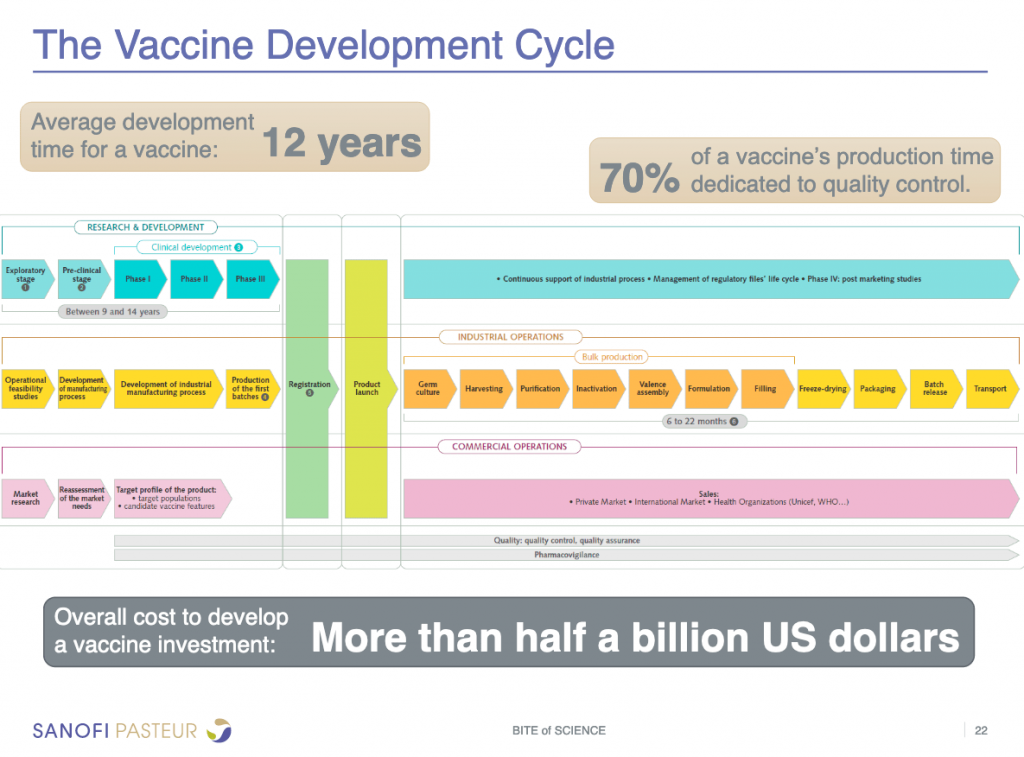
Developing a new replacement vaccine from scratch takes considerable time. It depends tons on what proportion information is out there about the disease itself, how the disease infects people and spreads, and so on. But it traditionally has taken 5-10 years to urge a replacement vaccine. that creates it truly amazing that we have already got one authorized vaccine for COVID-19, and are evaluating stage 3 clinical test data on others.
It speaks volumes about the efforts put into pandemic preparedness and response. As for actual manufacturing time, which will be suffering from the sort of vaccine being made – though this isn’t really relevant for COVID-19, since all of the vaccines being considered for COVID-19 take about an equivalent amount of your time to manufacture.
If we are talking a few vaccine that has already been tested and approved, we could generalize and say that one batch of vaccine, consisting of a few thousand doses, may take 2-6 weeks to travel from starting with raw materials to being a completed vaccine during a vial or syringe.
How long does it take to make conventional vaccines? And why does it take so long?
First, from the stand point of design, it takes a short time to know the disease, its path of infection and spread, so as to seek out how to prevent it. Also, once a thought to alert the system of the invader is conceived, then you would like to check the candidate to form sure the conceptual idea works. Once that’s acceptable, then you would like to determine a producing process that lends itself to large scale production… all those steps take time.
Second, from the stand point of producing , most vaccines are biologicals – meaning they’re produced with help from a microorganism. which means you would like time for the microorganisms to grow and obtain the work done.
Here’s a general overview of the process:To produce a vaccine employing a biological system, you initially select an appropriate host. this is often typically a well known organism like bacteria or yeast.
Then the genetic material of the host is engineered to supply instructions for the expression of the specified vaccine. In other words, you engineer the organism to form the vaccine for you.
The newly engineered organism is then grown in sufficient quantities to be used for production purposes.
Once expression is completed by the organism, our vaccine is separated from everything else the organisms produce using operations like filtration.
The last step is to combine the purified vaccine with the excipients –or those other components that add stability to the vaccine and permit us to securely transport and store it. The formulated vaccine is then filled into multi-dose vials or single-use syringes for administration.
And there’s another thing, once all the above is completed, then each batch produced must be tested for identity, purity and potency to form sure everyone receives a top quality product.
As you’ll imagine, all that takes weeks per batch.
What is mRNA Vaccines
Researchers are studying and dealing with mRNA vaccines for many years . Attention has grown in these vaccines because they will be advanced during a laboratory using willingly available materials. this suggests the method are often consistent and scaled up, making vaccine development faster than traditional methods of creating vaccines.
mRNA vaccines are studied before for flu, Zika, rabies, and cytomegalovirus (CMV). As soon because the required evidence about the virus that causes COVID-19 was available, scientists instigated designing the mRNA guidelines for cells to create the unique spike protein into an mRNA vaccine.
Future mRNA vaccine expertise may leave one vaccine to supply defence for several diseases, thus reducing the amount of shots needed for cover against common vaccine-preventable diseases.
Outside vaccines, cancer examine has used mRNA to spark the system to focus on specific cancer cells.
How COVID-19 mRNA Vaccines Work?
COVID-19 mRNA vaccines give directions for our cells to form a harmless piece of what’s called the “spike protein.” The spike protein is found on the surface of the virus that causes COVID-19.
COVID-19 mRNA vaccines are given within the upper arm muscle. Once the instructions (mRNA) are inside the immune cells, the cells use them to form the protein piece. After the protein piece is formed , the cell breaks down the instructions and gets obviate them.
Next, the cell shows the protein portion on its surface. Our immune systems identify that the protein doesn’t go there and start building an immune reaction and gathering antibodies, like what happens in natural infection against COVID-19.
At the top of the method , our bodies have learned the way to protect against future infection. The advantage of mRNA vaccines, like all vaccines, is those vaccinated gain this protection without ever having to risk the intense significances of getting sick with COVID-19.
How long does Covid vaccine last?
Studies have shown that the antibodies after COVID-19 vaccination will last for three months, but the upper limit is not yet known because these vaccines have only been administered for a few months. find more.
Routinely recommended vaccines
1914 – Pertussis vaccine
1926 – Diphtheria vaccine
1938 – Tetanus vaccine
1945 – Inactivated influenza vaccine – trivalent (shot; not routinely recommended)
1948 – Diphtheria, tetanus, and pertussis vaccines combined to form DTP
1955 – Inactivated polio vaccine (shot)
1962 – Live polio vaccine (oral)
1963 – Measles vaccine
1967 – Mumps vaccine
1969- Rubella vaccine
1971 – Measles, mumps and rubella vaccines combined to form MMR
1981 – Hepatitis B vaccine
1985 – Hib vaccine
1992 – DTaP vaccine
1995 – Varicella vaccine
1995-1996 – Hepatitis A vaccine
1998 – Rotavirus vaccine (Rotashield®)(removed from market 1999)
2000 – Pneumococcal conjugate vaccine
2003 – Intranasal influenza vaccine — trivalent (not recommended for use between 2016-2018)
2005 – Meningococcal (ACWY) conjugate vaccine for adolescents
2005 – Tdap vaccine for adolescents
2006 – HPV vaccine for adolescent girls (protected against 2 or 4 types of HPV)
2006 – Rotavirus vaccine (RotaTeq®)
2006 – Shingles vaccine (Zostavax® for adults 60 yrs & older)
2008 – Rotavirus vaccine (RotaRix®)
2009 – HPV vaccine for adolescent boys (version that protected against 4 types of HPV)
2013 – Inactivated and intranasal influenza vaccine — quadrivalent (the intranasal version was not recommended for use between 2016-2018)
2014 – Meningococcal B vaccine licensed
2014 – HPV vaccine that protects against 9 types of HPV (Gardasil® 9)
2017 – Second shingles vaccine (Shingrix® for adults 50 years & older)
2017 – Adult hepatitis B vaccine (Heplisav-B®)
Vaccines not routinely recommended
1798 – Smallpox vaccine (discontinued use for general population in US 1972)
1885 – First rabies vaccines
1896 – Typhoid fever vaccine
1927 – Tuberculosis vaccine (BCG)
1930s – Japanese Encephalitis (JE) vaccine
1935 – Yellow fever vaccine
1954 – Anthrax vaccine
1965 – Currently used anthrax vaccine
1986 – Currently used JE vaccine
1997 – Currently used rabies vaccines
1999-2002 – Lyme disease vaccine (no longer available)
2016-2018 – Intranasal influenza vaccine
Ref – www.chop.edu


I find the development and evolution of vaccines so interesting. To think that a vaccine can be developed so quickly is amazing! With the recent pandemic, I am learning more and more about science. I find the timeline you shared really interesting.
This is such an interesting subject for sure. Probably more important now than ever before with all what is going on at the moment
So interesting to read about all of this. I also was told the flu mist was ineffective all the years my kids took it. It will be interesting to see how the Covid vaccines go.
This was such an interesting read! It’s really amazing that although so many vaccines took decades (or over a century, in the case of the typhoid vaccine) to develop, the COVID-19 vaccines have been developed, tested, and are starting to be available in such a short time.
I trust and believe in science, so I understand how crucial vaccines are. It’s nice to learn more about the process.
Vaccines have been on my mind a lot lately, especially considering that both of my grown children and I are immunocompromised due to chronic illnesses (genetic). This was a fascinating read, as I did not really know anything about their development and history. I guess it is time to get more familiar with them. Blessings!
It’s so neat to learn about the process of vaccines. Now more than ever its important to understand this.
Very informative and apt topic to discuss now when we are hit so badly by the pandemic. A lot of experiments on with the vaccine and hope is that things will improve.
Vaccines are interesting. The ingredients in most of them are worth looking at too, like aluminum, mercury, formaldehyde, animal and human cells and more.
Thank goodness for science! I can’t wait to get my COVID vaccine <3. Thank you for sharing the story of vaccines today with us! Understanding vaccines is so important 🙂
Very informative and detailed post about vaccine and its history. Thank you very much for sharing this knowledge.
Yyyeeessssss….vaccines are something I need to know more of! It’s years of pain and tears making these substances that save our lives, on a daily.
It was an interesting read for me. I was amazed to read about its development and evolution!
Phentermine is the one that lets you have the best of support while on your way to losing fats.